| Title | Direct observation of multiple tautomers of oxythiamine and their recognition by the thiamine pyrophosphate riboswitch. |
| Publication Type | Journal Article |
| Year of Publication | 2014 |
| Authors | Singh V, Peng CSam, Li D, Mitra K, Silvestre KJ, Tokmakoff A, Essigmann JM |
| Journal | ACS Chem Biol |
| Volume | 9 |
| Issue | 1 |
| Pagination | 227-36 |
| Date Published | 2014 Jan 17 |
| ISSN | 1554-8937 |
| Keywords | Aptamers, Nucleotide, Isomerism, Oxythiamine, Riboswitch, Thiamine Pyrophosphate |
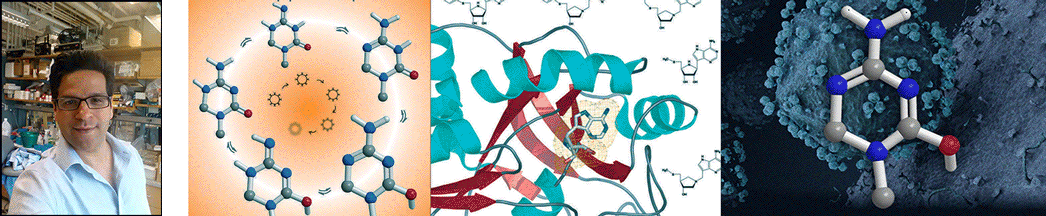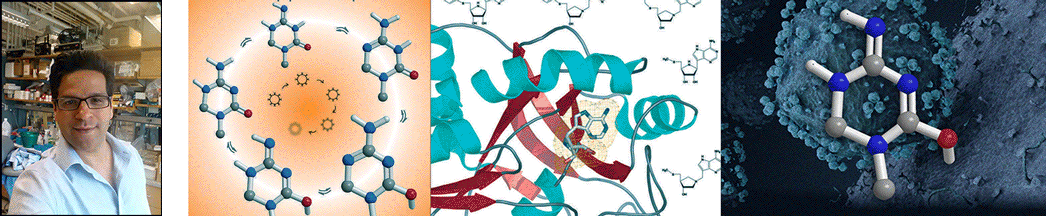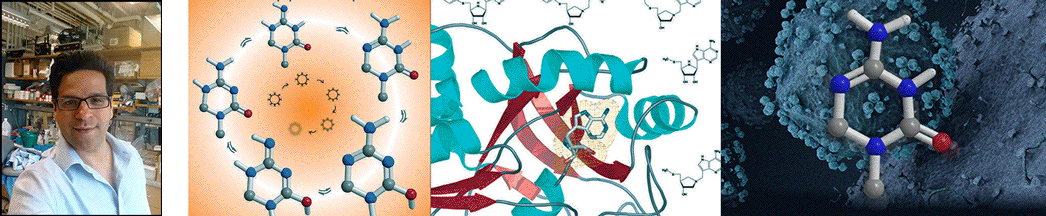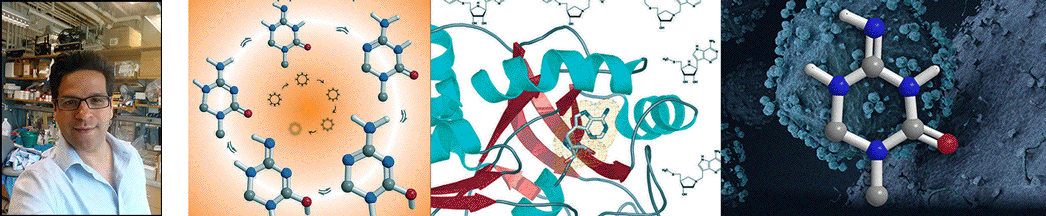
| Abstract | Structural diversification of canonical nucleic acid bases and nucleotide analogues by tautomerism has been proposed to be a powerful on/off switching mechanism allowing regulation of many biological processes mediated by RNA enzymes and aptamers. Despite the suspected biological importance of tautomerism, attempts to observe minor tautomeric forms in nucleic acid or hybrid nucleic acid-ligand complexes have met with challenges due to the lack of sensitive methods. Here, a combination of spectroscopic, biochemical, and computational tools probed tautomerism in the context of an RNA aptamer-ligand complex; studies involved a model ligand, oxythiamine pyrophosphate (OxyTPP), bound to the thiamine pyrophosphate (TPP) riboswitch (an RNA aptamer) as well as its unbound nonphosphorylated form, oxythiamine (OxyT). OxyTPP, similarly to canonical heteroaromatic nucleic acid bases, has a pyrimidine ring that forms hydrogen bonding interactions with the riboswitch. Tautomerism was established using two-dimensional infrared (2D IR) spectroscopy, variable temperature FTIR and NMR spectroscopies, binding isotope effects (BIEs), and computational methods. All three possible tautomers of OxyT, including the minor enol tautomer, were directly identified, and their distributions were quantitated. In the bound form, BIE data suggested that OxyTPP existed as a 4'-keto tautomer that was likely protonated at the N1'-position. These results also provide a mechanistic framework for understanding the activation of riboswitch in response to deamination of the active form of vitamin B1 (or TPP). The combination of methods reported here revealing the fine details of tautomerism can be applied to other systems where the importance of tautomerism is suspected. |
| DOI | 10.1021/cb400581f |
| Alternate Journal | ACS Chem Biol |
| PubMed ID | 24252063 |
| PubMed Central ID | PMC3956446 |
| Grant List | CA26731 / CA / NCI NIH HHS / United States T32 ES007020 / ES / NIEHS NIH HHS / United States P30 ES002109 / ES / NIEHS NIH HHS / United States ES002109 / ES / NIEHS NIH HHS / United States P01 CA026731 / CA / NCI NIH HHS / United States R01 CA080024 / CA / NCI NIH HHS / United States Y02 ES007020 / ES / NIEHS NIH HHS / United States P41-EB015871 / EB / NIBIB NIH HHS / United States P41 EB015871 / EB / NIBIB NIH HHS / United States P30-ES002109 / ES / NIEHS NIH HHS / United States CA080024 / CA / NCI NIH HHS / United States R37 CA080024 / CA / NCI NIH HHS / United States |
Direct observation of multiple tautomers of oxythiamine and their recognition by the thiamine pyrophosphate riboswitch.
Submitted by Vipender Singh on